Latest Developments
These results provide further evidence that a three-dose vaccine regimen benefits the induction of optimal functional T cell immune memory.
 |
| Table 1. Vaccines do generate populations of highly-specific T cells (Source @erictopol) |
Innate Immune System vs Adaptive Immune System
Antibody-Mediated Immunity (or Humoral Immunity)
- Involves substances found in the humors (or body fluids)
- The aspect of immunity that is mediated by macromolecules found in extracellular fluids such as:
- Secreted antibodies
- Complement proteins
- Certain antimicrobial peptides
- Contrasts with cell-mediated immunity
Immune Memory
A thorough understanding of immune memory to SARS-CoV-2 requires evaluation of its various components, including:[2]
- Antigen Presenting Cells (APCs which includes macrophages, dendritic cells, B cells)
- In the steady state, and when the body is challenged by injury and infection, dendritic cells (one type of APCs) travel from body surfaces to immune or lymphoid tissues, where they home to regions rich in T cells. There, dendritic cells deliver two types of information:
- they display antigens, the substances that are recognized by T cells,
- they alert these lymphocytes to the presence of injury or infection.
- B Cells (aka B lymphocytes)
- Function in the humoral immunity component of the adaptive immune system by secreting antibodies
- Present antigens and secrete cytokines
- Express B cell receptors (BCRs) on their cell membrane.
- BCRs allow the B cell to bind to a specific antigen, against which it will initiate an antibody response.
- CD8+ T Cells (aka killer T-cells or cytotoxic T cells)
- Are T lymphocytes that kill cancer cells, cells that are infected (particularly with viruses), or cells that are damaged in other ways
- Most cytotoxic T cells express T-cell receptors (TCRs) that can recognize a specific antigen
- CD4+ T Cells (i.e., T helper cells)
- Help the activity of other immune cells by releasing cytokines, small protein mediators that alter the behavior of target cells that express receptors for those cytokines.
- Help to polarize the immune response into the appropriate kind depending on the nature of the immunological insult (virus vs. extracellular bacterium vs. intracellular bacterium vs. helminth vs. fungus vs. protist).
- Are essential in B cell antibody class switching, breaking cross-tolerance in dendritic cells, in the activation and growth of cytotoxic T cells, and in maximizing bactericidal activity of phagocytes such as macrophages and neutrophils.
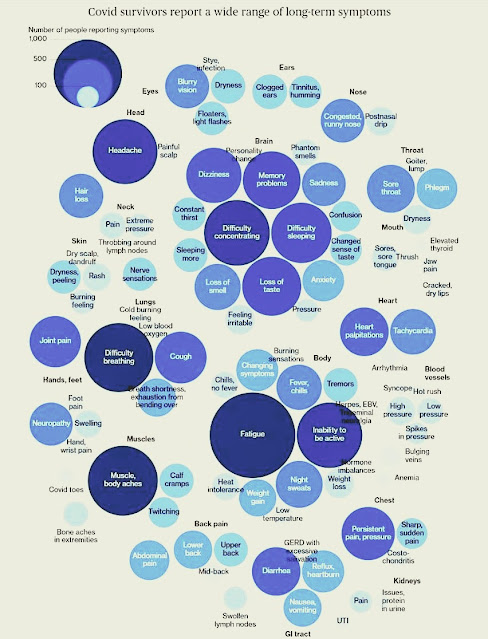
A Cross-Sectional Study
- Re-infection with SARS-CoV-2
- 2° COVID-19 disease
- B cells
- Overall, based on the observations, development of B cell memory to SARS-CoV-2 appeared to be robust and likely long-lasting
- CD8+ T cells
- The memory CD8+ T cell half-lives (or t1/2) observed herein were comparable to the 123d t1/2 observed for memory CD8+ T cells within 1-2 years after yellow fever immunization.[10]
- Overall, the decay of circulating SARS-CoV-2-specific CD8+ T cell is consistent with what has been reported for another acute virus.
- CD4+ T cells
- Circulating SARS-CoV-2 memory CD4+ T cell responses were quite robust
- 94% of subjects with detectable circulating SARS-CoV-2 memory CD4+ T cells at 1 month PSO
- 89% of subjects with detectable circulating SARS-CoV-2 memory CD4+ T cells at≥ 6 months PSO
Hybrid Immunity
Hybrid vigor can occur when different plant lines are bred together and the hybrid is a much stronger plant. Something similar happens when natural immunity is combined with vaccine-generated immunity, resulting in 25 to 100 times higher antibody responses, driven by memory B cells and CD4+ T cells and broader cross-protection from variants.
- To produce identical antibodies upon reinfection with the same virus
- To encode a library of antibody mutations, a stock-pile of immunological variants
A substantial proportion of memory B cells encode antibodies that are capable of binding or neutralizing VOCs, and the quality of these memory B cells increase over time. Thus the increase in variant-neutralizing antibodies after vaccination of previously SARS-CoV-2-infected persons reflects recall of diverse and high-quality memory B cells generated after the original infection.
References
- Human T Cell Memory: A Dynamic View
- Immunological memory to SARS-CoV-2 assessed for greater than six months after infection
- W. A. Orenstein, R. Ahmed, Simply put: Vaccination saves lives. Proc National Acad Sci. 114, 4031–4033 (2017).
- P. Piot, H. J. Larson, K. L. O’Brien, J. N’kengasong, E. Ng, S. Sow, B. Kampmann, Immunization: vital progress, unfinished agenda. Nature. 575, 119–129 (2019).
- S. Plotkin, W. Orenstein, P. Offit, Plotkin’s vaccines, 7th edition (Elsevier, 2018), Elsevier.
- D. S. Stephens, M. J. McElrath, COVID-19 and the Path to Immunity. Jama. 324 (2020), doi:10.1001/jama.2020.16656.
- F. Krammer, SARS-CoV-2 vaccines in development. Nature, 1–16 (2020).
- S. M. Kissler, C. Tedijanto, E. Goldstein, Y. H. Grad, M. Lipsitch, Projecting the transmission dynamics of SARS-CoV-2 through the postpandemic period. Science. 368, 860–868 (2020).
- C. M. Saad-Roy, C. E. Wagner, R. E. Baker, S. E. Morris, J. Farrar, A. L. Graham, S. A. Levin, M. J. Mina, C. J. E. Metcalf, B. T. Grenfell, Immune life history, vaccination, and the dynamics of SARS-CoV-2 over the next 5 years. Science, eabd7343 (2020).
- R. S. Akondy, M. Fitch, S. Edupuganti, S. Yang, H. T. Kissick, K. W. Li, B. A. Youngblood, H. A. Abdelsamed, D. J. McGuire, K. W. Cohen, G. Alexe, S. Nagar, M. M. McCausland, S. Gupta, P. Tata, W. N. Haining, M. J. McElrath, D. Zhang, B. Hu, W. J. Greenleaf, J. J. Goronzy, M. J. Mulligan, M. Hellerstein, R. Ahmed, Origin and differentiation of human memory CD8 T cells after vaccination. Nature. 552, 362–367 (2017).
- Vaccine bootcamp (nice animation)
- Human Coronavirus: Host-Pathogen Interaction
- Hybrid Immunity
- Understanding the Basics of Memory B Cells—The Antibody Factory

- Antigen-Presenting Cells
- Resilient T cell responses to B.1.1.529 (Omicron) SARS-CoV-2 variant




![Understanding Asthma [CHART]](https://www.shape-able.com/wp-content/uploads/Understanding-Asthma-Chart.jpg)